Introduction: Circulating clonal plasma cell (cCPC) is an easily obtained and non-invasive biomarker in multiple myeloma (MM), providing distinct prognostic information from the revised-international staging system (R-ISS). However, its prognostic relevance has been investigated primarily in transplant-eligible and clinical trial settings. Consequently, there is limited data on the impact of cCPC on outcomes and optimal cut-off values in transplant-ineligible (TIE) MM patients. Moreover, appropriate combinations to refine risk stratification, such as tumor burden measured by positron emission tomography-computed tomography (PET-CT), have yet to be fully explored.
Methods: We performed a retrospective analysis on newly diagnosed TIE-MM patients with available peripheral blood (PB) flow cytometry (FCM) and PET-CT data from 2011 to 2021. Patients with primary plasma cell leukemia (n=8), defined as >5% monoclonal plasma cells in PB, were excluded from the study. Quantification of cCPC was performed using 6-color (2011-2016) or 8-color (2017-2021) FCM staining for CD19, CD56, CD38, CD138, cytoplasmic κ, and cytoplasmic λ to confirm monoclonality (Beckman Coulter, USA), which required at least 150000 mononuclear cell counts with the detectable limit at 0.01% (10 -4) based on previous reports (Gonsalves et al). Tumor burden was assessed by calculating total lesion glycolysis (TLG) using the following formula: the mean standardized uptake value (SUV) multiplied by the total tumor volume with an absolute SUV >2.5, using Metavol Software (Hokkaido University, Japan). Receiver operating curve analysis with the Youden index was employed to determine the optimal cutoff for predicting 2-year progression-free survival (PFS) and 5-year overall survival (OS). The predictive performance of R-ISS, cCPC, and TLG alone, along with their combinations, was compared using Harrell's concordance index (c-index).
Results: 114 patients with TIE-MM were analyzed. The median age was 78 years (interquartile range [IQR] 73-83). The number of patients classified as R-ISS Ⅰ, Ⅱ, and Ⅲ was 12 (10.5%), 82 (71.9%), and 20 (17.5%), respectively. Most patients (85.1%) received proteasome inhibitor-based induction remission therapy.
cCPCs were detectable in 90 patients (80.7%), with a median level of 6 ×10 -4 (IQR 1-22). Regarding tumor burden, 73 patients (64%) had at least one fluorodeoxyglucose avid lesion with a median TLG of 34.7 (IQR 0-268). cCPC levels were significantly correlated with CPC in bone marrow (R =0.24, P =0.01) but not with TLG (R =-0.01, P =0.88) and involved free light chain levels (R =0.07, P =0.41).
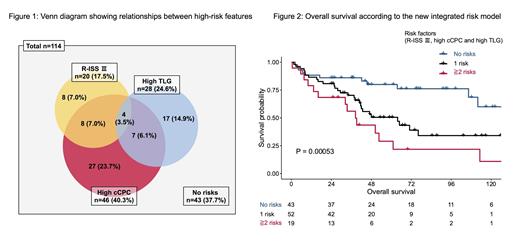
The optimal cut-off values in predicting 2-year PFS and 5-year OS were 9 and 10 ×10 -4 for cCPC, and 325 and 343 for TLG, respectively. For simplicity, we classified >10 ×10 -4 for cCPC and >300 for TLG as high-risks. Notably, approximately two-thirds of the patients (62.3%) had at least one high-risk feature (Figure 1). Both high cCPC and TLG showed significant associations with worse OS and PFS (5-year OS rate: cCPC high vs. low, 40.2% vs. 69.3%; TLG high vs. low, 38% vs. 64.5%; 2-year PFS rate: cCPC high vs. low, 45.7% vs. 69.1%; TLG high vs. low, 46.4% vs. 64% all P < 0.05). These factors retained prognostic significance after adjustment for R-ISS on multivariate analysis.
Finally, we constructed a novel predictive model integrating RISS, cCPC, and TLG. This model discriminated OS among patients with 0 (5-year OS rate, 80% [95% confidence interval 68.4%-93.6%]), 1 (50.5% [37.7%-67.7%]), and ≧2 factors (29% [13.3%-63.5%], P <0.001, Figure 2). The c-index was the highest (0.651) in this combined model compared to the single use of these risk factors (0.585, 0.594, and 0.569).
Conclusions: We demonstrated the prognostic impact of cCPC, which is independently associated with R-ISS and TLG, on real-world TIE-MM patients. The new prediction model, integrated with these different clinical and biological features, may provide robust outcome discrimination.
Disclosures
Matsue:AstraZeneca: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal